|
False Positivesġ1 of the 1,877 patients tested were deemed to be false positives, defined as being MOG-IgG seropositive with a titer of ≥1:20 but having a clinical syndrome inconsistent with MOG-AD and/or a more likely alternative diagnosis. Only one non-neurologist ordered a MOG-IgG test. The PPV at MOG-IgG ≥1:20 was robust whether the test was ordered by a neuroimmunologist (PPV of 82%) or neurologist without neuroimmunology expertise (PPV of 94%) (P=0.17 for the comparison). Using a MOG-IgG titer cutoff of ≥1:40, PPV improved to 92.3%. 
Using a serum MOG-IgG titer cutoff of ≥1:20, 67 of the 78 patients with positive MOG-IgG1 results were determined to be true positives. 
PPVs were calculated by dividing the number of true positives by all positive cases. Patients who were seropositive for MOG-IgG had clinical features of MOG-AD, and that had no more likely alternative diagnoses were considered true positives. The records of 78 patients who had positive results were retrospectively reviewed by two neurologists for typical MOG-AD clinical features. The researchers identified 1,877 patients who underwent serum MOG-IgG testing with the Mayo assay at the Brigham or Mass General between January 1, 2017, and March 25, 2021. They discuss cutoff titers in Frontiers in Neurology. Manzano, MD, a neuroimmunologist and neurohospitalist in the Department of Neurology at Brigham and Women’s Hospital and Massachusetts general Hospital, Marcelo Matiello, MD, a member of the division of neuro-immunology and comprehensive neurology at Massachusetts General Hospital, and colleagues determined positive predictive values (PPVs) of the assay in a real-world cohort. A live cell–based serum assay (Mayo Laboratories, Rochester, MN) is now available to detect MOG–immunoglobulin G1 (MOG-IgG1), the diagnostic biomarker for MOG-AD. The purpose of this study is to demonstrate the clinical efficacy of rozanolixizumab in maintenance treatment and asses safety and tolerability of rozanolixizumab in adult study participants with primary immune thrombocytopenia (ITP).Myelin oligodendrocyte glycoprotein antibody–associated disease (MOG-AD), a central nervous system (CNS) demyelinating disease, can present with acute disseminated encephalomyelitis, optic neuritis, and/or transverse myelitis. 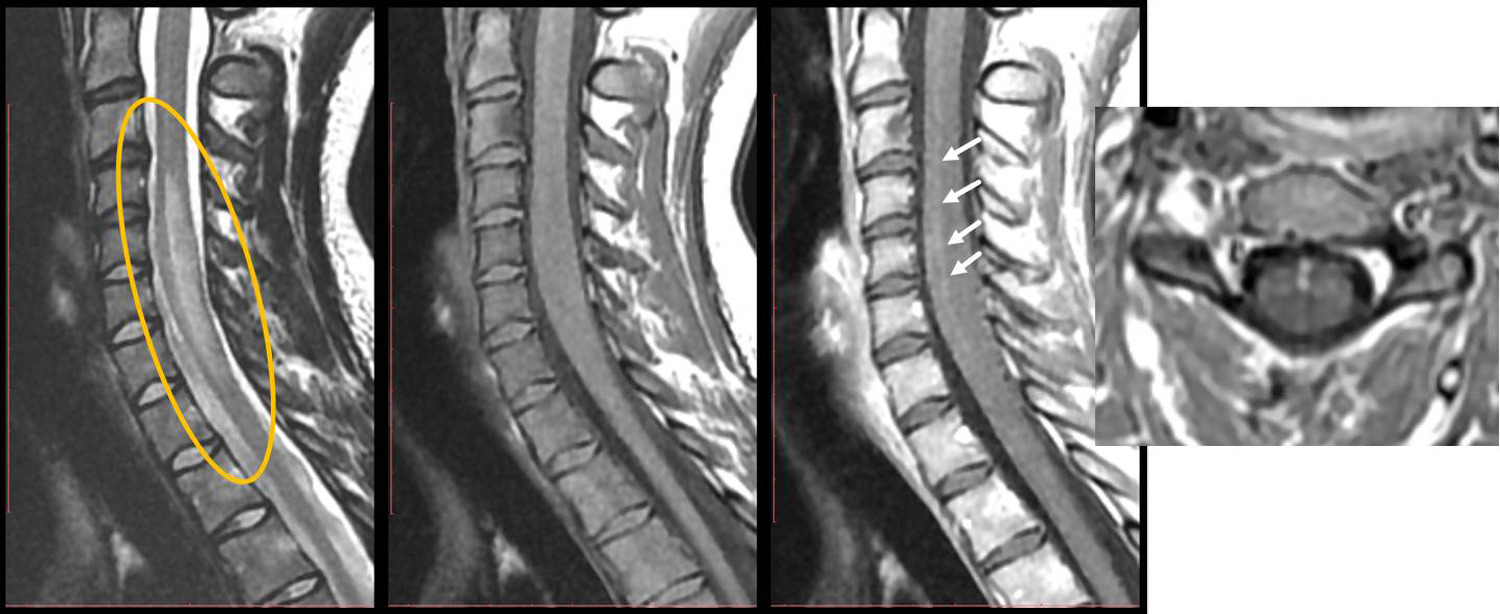
Myelin oligodendrocyte glycoprotein antibody-associated diseaseĪ Study to Evaluate the Efficacy and Safety of Rozanolixizumab in Adult Participants With Myelin Oligodendrocyte Glycoprotein (MOG) Antibody-associated Disease (MOG-AD) (cosMOG) Robak T., Blood advances 2020 4(17):4136–46Ī Study to Assess Long-term Safety, Tolerability and Efficacy of Rozanolixizumab in Subjects With Chronic Inflammatory Demyelinating PolyradiculoneuropathyĪ Study to Investigate the Long-term Safety, Tolerability, and Efficacy of Rozanolixizumab in Adult Patients With Generalized Myasthenia GravisĪ Study to Evaluate Rozanolixizumab in Study Participants With Generalized Myasthenia GravisĪ Phase 3, Open-label, Crossover Study to Evaluate Self-administration of Rozanolixizumab by Study Participants With Generalized Myasthenia Gravis (gMG) Study to Evaluate Safety, Tolerability and Efficacy of UCB7665 in Subjects With Primary Immune Thrombocytopenia , J Periph Nerv Syst 2022 27(S3 suppl.):S110-111, 2022 Peripheral Nerve Society Annual Meeting, May 14-17, 2022 Miami, Fl, USA Querol L., de Seze J., Dysgaard T., Levine T., Rao T.H. , Neurology 2022 99(23 suppl.2):S42-S43, AAN 2022Ĭhronic Inflammatory Demyelinating PolyradiculoneuropathyĪ Study to Assess the Efficacy, Safety and Tolerability of Rozanolixizumab in Subjects With Chronic Inflammatory Demyelinating Polyradiculoneuropathy
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
